Association between Spirometric Parameters and Depressive Symptoms in New Mexico Uranium Workers
 Saturday, February 13, 2021 at 8:00AM
Saturday, February 13, 2021 at 8:00AM Shiva Sharma MD, MPH1
Xin W. Shore MS2
Satyajit Mohite MD, MPH3
Orrin Myers PhD2
Denece Kesler MD, MPH1
Kevin Vlahovich MD, MS1
Akshay Sood MD, MPH4
1Preventive Medicine Section, Department of Internal Medicine, University of New Mexico School of Medicine, Albuquerque, NM USA
2Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque, NM USA
3Department of Behavioral Health, Psychiatry & Psychology, Mayo Clinic Health System, Mankato, MN USA
4Division of Pulmonary, Critical Care and Sleep Medicine, Department of Internal Medicine, University of New Mexico School of Medicine, Albuquerque, NM USA
Abstract
Background: Uranium workers are at risk of developing lung disease, characterized by low forced expiratory volume in one second (FEV1) and/or forced vital capacity (FVC). Previous studies have found an association between decreased lung function and depressive symptoms in patients with pulmonary pathologies, but this association has not been well examined in occupational cohorts, especially uranium workers.
Methods: This cross-sectional study evaluated the association between spirometric measures and depressive symptoms in a sample of elderly former uranium workers screened by the New Mexico Radiation Exposure Screening & Education Program (NM-RESEP). Race- and ethnicity-specific reference equations were used to determine predicted spirometric indices (predictor variable). At least one depressive symptom [depressed mood and/or anhedonia, as determined by a modified Patient Health Questionnaire-2 (PHQ-2)], was the outcome variables. Chi-square tests and multivariable logistic regression models were used for statistical analyses.
Results: At least one depressive symptom was self-reported by 7.6% of uranium workers. Depressed mood was reported over twice as much as anhedonia (7.2% versus 3.3%). Abnormal FVC was associated with at least one depressive symptom after adjustment for covariates. There was no significant interaction between race/ethnicity and spirometric indices on depressive symptoms.
Conclusions: Although depressive symptoms are uncommonly reported in uranium workers, they are an important comorbidity due to their overall clinical impact. Abnormal FVC was associated with depressive symptoms. Race/ethnicity was not found to be an effect modifier for the association between abnormal FVC and depressive symptoms. To better understand the mechanism underlying this association and determine if a causal relationship exists between spirometric indices and depressive symptoms in occupational populations at risk for developing lung disease, larger longitudinal studies are required. We recommend screening for depressive symptoms for current and former uranium workers as part of routine health surveillance of this occupational cohort. Such screening may help overcome workers’ reluctance to self-report and seek treatment for depression and may avoid negative consequences to health and safety from missed diagnoses.
Introduction
Uranium workers are at risk of pulmonary injury via two primary mechanisms: inhalation of radon daughters causing radiation-induced lung damage (1,2) and dust inhalation (3). Exposed workers are additionally at risk for developing cardiovascular pathology (4). Lung diseases can result in a clinically significant decline in pulmonary function and have been associated with various neuropsychiatric sequelae (5,6). Screening for and treatment of depression in interstitial lung disease (ILD) has been proposed to improve quality of life (6-8). Significant levels of depressive symptoms are described in patients with silicosis (8) and may adversely affect quality of life (9). In a study of patients with ILD, depressive symptoms correlate with dyspnea, forced vital capacity (FVC), sleep quality, and pain (7).
Presence of depressed mood or anhedonia, which is a significant decrease in deriving pleasure from the majority of one’s daily activities, on most days, is requisite for diagnosis of major depressive disorder (10). Individual inquiry of depressed mood has demonstrated 85-90% sensitivity for detection of depression; and addition of another question, specific for anhedonia, raises overall sensitivity to 95% for the two-question inquiry (11).
Our objective was to evaluate the prevalence of depressive symptoms in uranium workers, to examine their association with spirometric values, and examine race/ethnicity interaction with spirometry on depressive symptoms. We studied uranium workers enrolled in the New Mexico Radiation Exposure Screening and Education Program (NM-RESEP). New Mexico workers have been significantly impacted by uranium extraction activities and many are compensated through the Radiation Exposure Compensation Act (RECA) (12). The findings from this study may help elucidate the biopsychosocial impact of uranium-related lung abnormalities in New Mexico workers.
Methods
Study Design:
This is a cross-sectional analysis of baseline evaluation data from former New Mexico uranium workers (i.e., miners, millers, and ore transporters) voluntarily enrolled between 2004 and 2017 in NM-RESEP, a federally-funded health-screening and education program, located at the University of New Mexico (UNM) Health Sciences Center, serving Albuquerque and surrounding communities.
Data Collection:
Data were obtained from a self-reported questionnaire administered by a trained interviewer and confirmed by a physician/nurse practitioner. The questionnaire included demographics, severity of dyspnea via the modified Medical Research Council (mMRC) Dyspnea Scale (13), information on smoking history, cardiovascular status, and screening for depressive symptoms (using the modified Patient Health Questionnaire or PHQ-2) which includes two items on depressed mood and anhedonia (14). Body mass index (BMI) was calculated using measured height and weight. A prebronchodilator spirometry was obtained by trained technicians, utilizing standard guidelines from the American Thoracic Society and the European Respiratory Society (15). Test results were independently reviewed for quality by a pulmonologist. Gender- and race/ethnicity- specific reference equations were used to determine predicted normative values for spirometry (16). Abnormal values were defined by the lower limit of normal obtained from reference standards. Data was entered into a secure web-based Research Electronic Data Capture (REDCap) database.
Predictor and Outcome Variables:
The outcome included a positive response for PHQ-2 item on either depressive symptom, i.e., depressed mood or anhedonia. Predictor variables included spirometric parameters and race/ethnicity, including the absolute and percent-predicted values for FVC), forced expiratory volume in one second (FEV1), and the absolute value of the FEV1/FVC ratio. Lower limits of normal obtained from the reference standards from the Third National Health and Nutrition Examination Survey (NHANES III) were used to define abnormal spirometric values (16).
Statistical Methods:
Frequencies, percentages, means, and standard deviations in a univariate analysis were reported. For the purpose of analyses, the outcome variable was endorsement of either depressive symptom. Chi-Square tests were used to analyze categorical outcome variables and generate p-values to determine significance of the findings. In the multivariable logistic regression analysis, variables that were evaluated for potential confounding included smoking status and pack-years.
Ethical Approval and Funding:
This study was approved by the UNM Institutional Review Board or Human Resources Protections Office (14-058). The study was supported by NM-RESEP, which is funded by the Health Resources Services Administration (HRSA), and UNM Health Science Center CTSC Grant Number: UL1TR001449.
Results
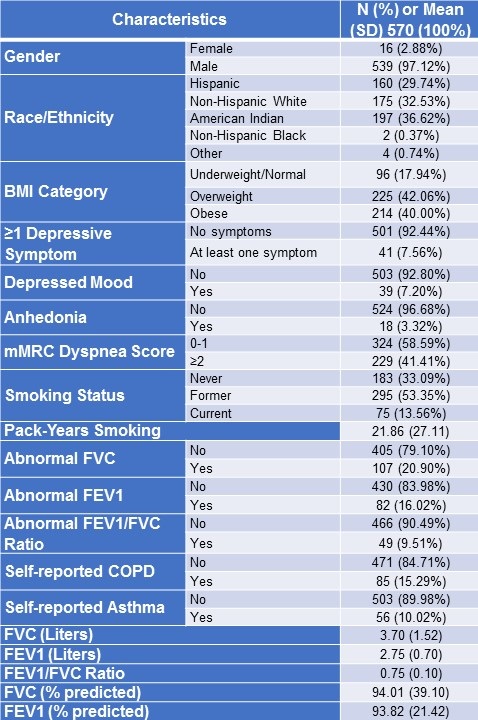
Subject characteristics are shown in Table 1. Of the 570 uranium workers, 97.1% were men, 66.7% were of racial/ethnic minority with the largest group being American Indian (36.6%). Most workers were older (mean age of 68.5 ± 8.1 years) with BMI values in the overweight or obese categories (82.1%). 7.6% of workers reported at least one depressive symptom, with 7.2% and 3.3% reporting depressed mood and anhedonia, respectively. The prevalence of at least one depressive symptom in Hispanic, American Indian, and non-Hispanic White workers were 11.4%, 7.2%, and 4.1%, respectively (p=0.14 for all race/ethnicity group comparison) and post-hoc comparison between Hispanic and non-Hispanic White workers was significant (p=0.001) (not shown in Table 1). 66.9% of workers were either former or current smokers. With regards to previous pulmonary history, 15.3% and 10.0% of workers reported positive history of COPD and asthma, respectively.
Table 1. NM-RESEP Uranium Workers (2004-2017).
Both unadjusted univariate and adjusted multivariable analyses revealed that workers with abnormal FVC were at least 2.9 times more likely to endorse at least one depressive symptom. No associations were found between abnormal FEV1 or abnormal FEV1/FVC ratios and depressive symptoms (Table 2).
Table 2. Unadjusted and Adjusted Associations of the Presence of Depressive Symptoms on Spirometric Indices.
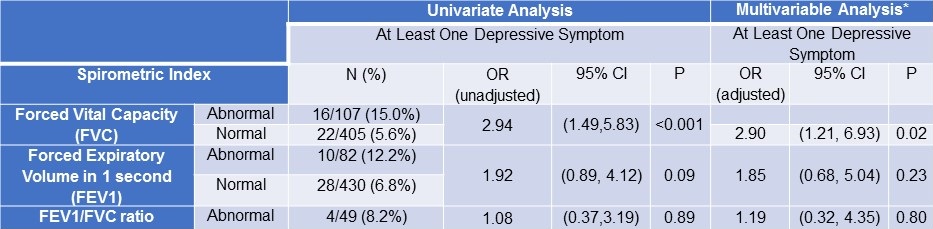
*Covariates in the above multivariable model using logistic regression analysis included: smoking status and smoking pack-years. **Further adjustment for the following covariates: age, gender, and race/ethnicity did not change results in the multivariable model (FVC OR: 2.86, 95% CI: 1.18-6.96, p=0.02).
Although the associations between spirometric indices and depressive symptoms appeared stronger among Hispanic workers than other race/ethnicity subgroups, this was not borne by a formal test of interaction between race/ethnicity and spirometric indices on either depressive symptom. However, interaction testing identified a trend towards significance for Hispanic workers between abnormal FEV1 and self-reporting of depressive symptoms (p=0.07) (Table 3).
Table 3: Interaction between Spirometric Indices and Race/Ethnicity on Depressive Symptoms.
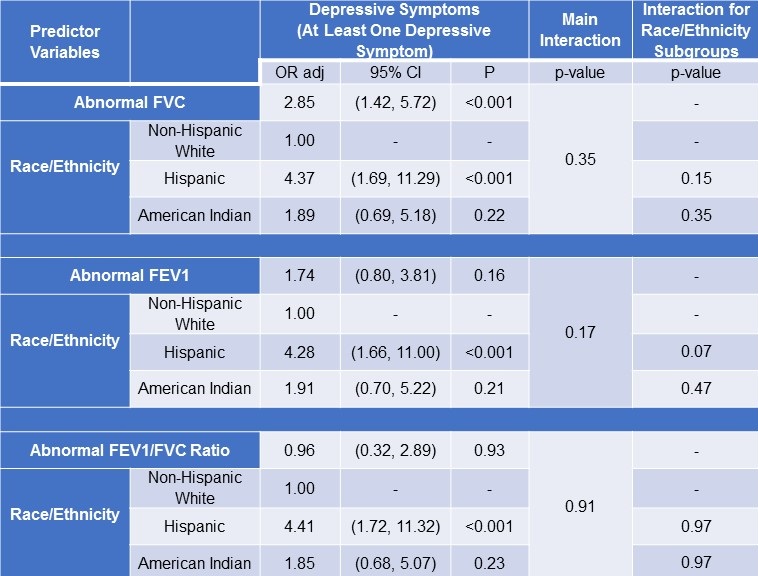
*Logistic regression analysis was used.
Discussion
A minority of uranium workers sampled in this secondary analysis self-reported at least one depressive symptom (7.6%). Depressed mood was reported over twice as much as anhedonia was reported (7.2% vs 3.3%). Abnormal FVC on spirometry was found to be associated with depressive symptoms after adjustment for covariates. There was no significant interaction between race/ethnicity and spirometric indices on depressive symptoms.
Uranium ore extraction in New Mexico occurs in open pit or underground mines. Subsequently, uranium is isolated from ore via milling or heap leaching (17). Most of the workers in this study were subjected to hazardous working conditions marked by lack of provision of personal protective equipment (including respirators) to handle uranium and inhalational dust exposure. Inadequate ventilation in underground mines also led to increased radon and dust exposure and workers were not adequately informed of these occupational exposures by mining companies or federal agencies (i.e. US Atomic Energy Commission, Nuclear Regulatory Commission, US Department of Energy) (12).
Uranium enters the human body primarily via inhalation and ingestion (18). It deposits primarily in the lungs and skeleton (insoluble uranium) and kidneys (soluble uranium) (19) where it causes chemical and radiological damage to these organs (20). In a murine study, uranium was found to enter the central nervous system, crossing the blood-brain barrier and accumulating in the hippocampus, resulting in detrimental neurophysiological effects and changes in REM sleep patterns (21). A case study involving 81 American Indian uranium workers found anxiety and depression to be the most common mental health problems, and respiratory complaints and skin rashes were the most common physical health issues (22). Radon gas, a byproduct of the uranium decay process, attaches to dust particles and when inhaled into the lungs the alpha radiation released by radon daughters damages lung tissue. Like non-uranium industry workers engaged in other types of mining-related activities, uranium workers are at risk for occupational pneumoconiosis, presenting with features similar to silicosis (23), and chronic fibrotic ILD (3). Pneumoconiosis has been associated with increased risk of other pulmonary conditions, including pulmonary emboli (24), lung carcinoma (23), chronic obstructive pulmonary disease (COPD) (26), tuberculosis (27), and clinically significant decline in lung function (28).
Many chronic pulmonary conditions such as asthma, COPD (29), bronchiectasis (30), and lung cancer (31) are associated with depressive symptoms. In a prospective study of patients with bronchiectasis, low FEV1 values were observed among patients with depressive symptoms (30). In a study of French dairy farmers, Guillien (32) found depression was associated with lower FEV1. A 2013 systematic review and meta-analysis revealed that the relationship between COPD and depression is bidirectional (33). Our study did not contain information regarding history of a prior or current diagnosis of depression for enrolled patients, thus our secondary analysis is not a like-for-like comparison to existing literature on this topic. Our study involved individuals with mostly normal lung function, indicating that the association between abnormal FVC and depressive symptoms may be seen relatively early in the disease course.
Psychosocial factors may play a role in the development of workplace-associated disability in workers with respiratory impairment, but evidence-based guidance to address these psychosocial factors is limited (34). The low prevalence of depressive symptoms in our study may reflect the high proportion of men enrolled (97.1%), as overall prevalence of depression in men is approximately half that of women (35). Alternatively, men may under-report due to a lack of awareness and understanding of depression and fear of stigmatization for self-reporting amongst coworkers or wider society. Additionally, use of the standard PHQ-2 and DSM diagnostic criteria in American Indians may not produce reliable results due to potential cultural and linguistic differences (36). The rate of depression in American indigenous populations has found to be 8.9% (which is higher than all other racial/ethnic groups except biracial individuals) and can range from 10-30% (37), however, the prevalence of depressive symptoms in American Indian workers in our study was 7.2%. To the best of our knowledge, no validation studies have been performed for use of any version of the PHQ-2 in New Mexican American Indian populations. The PHQ-2 has been validated in English- and Spanish-speaking Hispanic Americans (38). Perini found ethnic minorities diagnosed with “chronic nonspecific lung disease” exhibited higher absolute prevalence of depressive symptoms than the ethnic majority (29). Our study findings partially agree with Perini’s findings in that Hispanic uranium workers were more likely to endorse depressed mood than non-Hispanic White workers.
Our study was a cross-sectional, secondary analysis of an occupational cohort of mostly elderly, former uranium workers enrolled in NM-RESEP. Longitudinal analysis of this association may further elucidate the direction of the association. Our study could benefit from culture-specific depression diagnostic criteria paired with spirometric measures to specific pulmonary diagnoses. While anhedonia has customarily been associated with loss of pleasure (10), the construct has recently expanded to include interest in activity, effort, and discrimination between anticipation and consummatory forms of pleasure. New approaches for anhedonia assessment are in development (39). Our assessment of anhedonia may have been limited and a more robust screening tool that screens for additional depressive symptoms beyond depressed mood and anhedonia, such as the PHQ-9 or Hospital Anxiety and Depression Scale (HADS) rather than the PHQ-2, could improve result validity. As depression has a complex nature, a more rigorous biopsychosocial assessment would help in determining the role pulmonary pathology plays in depression in this study sample. To the best of our knowledge, this is the first study to examine the association of spirometric indices with depressive symptoms in former uranium workers. The strengths of our study include the robust participation of minority workers due to use of a mobile screening unit, its clinical relevance in light of ongoing uranium-associated activity, and potential future impact on health. Further study on this topic is merited as untreated depression in workers poses potential risks to workplace safety. As industrial use of nuclear material continues in the United States and other countries such as Kazakhstan, Canada, and Australia, this area of study is relevant to occupational health on a global scale. We recommend screening for depressive symptoms in current and former uranium workers as part of routine health surveillance to better address reluctance to self-report and seek treatment for depression, as well as to avoid potential negative consequences to health and safety from a missed diagnosis.
Acknowledgments
Guarantor: Akshay Sood MD, MPH takes responsibility for the content of the manuscript, including the data and analysis.
Author contributions: All authors had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. All authors contributed substantially to the data analysis and interpretation and the writing of the manuscript.
Financial/non-financial disclosures: All authors report no conflict of interest.
Abbreviation List
- BMI: body mass index
- COPD: chronic obstructive pulmonary disease
- CTSC: Clinical and Translational Science Center
- DSM: Diagnostic and Statistical Manual of Mental Disorders
- FEV1: forced expiratory volume in one second
- FVC: forced vital capacity
- HADS: Hospital Anxiety and Depression Scale
- ILD: interstitial lung disease
- mMRC: modified Medical Research Council
- NHANES III: The Third National Health and Nutrition Examination Survey
- NM-RESEP: New Mexico Radiation Exposure Screening and Education Program
- PHQ-2/PHQ-9: Patient Health Questionnaire-2/Patient Health Questionnaire-9
- RECA: Radiation Exposure Compensation Act
- REDCap: Research Electronic Data Capture
- UNM: University of New Mexico
References
- Bersimbaev RI, Bulgakova O. The health effects of radon and uranium on the population of Kazakhstan. Genes Environ. 2015 Oct 1;37:18. [CrossRef] [PubMed]
- Faa A, Gerosa C, Fanni D, Floris G, Eyken PV, Lachowicz JI, Nurchi VM. Depleted Uranium and Human Health. Curr Med Chem. 2018;25(1):49-64. [CrossRef] [PubMed]
- Yen CM, Lin CL, Lin MC, Chen HY, Lu NH, Kao CH. Pneumoconiosis increases the risk of congestive heart failure: A nationwide population-based cohort study. Medicine (Baltimore). 2016 Jun;95(25):e3972. [CrossRef] [PubMed]
- Al Rashida VJM, Wang X, Myers OB, Boyce TW, Kocher E, Moreno M, Karr R, Ass'ad N, Cook LS, Sood A. Greater Odds for Angina in Uranium Miners Than Nonuranium Miners in New Mexico. J Occup Environ Med. 2019 Jan;61(1):1-7. [CrossRef] [PubMed]
- Dodd JW. Lung disease as a determinant of cognitive decline and dementia. Alzheimers Res Ther. 2015 Mar 21;7(1):32. [CrossRef] [PubMed]
- Ryerson CJ, Arean PA, Berkeley J, Carrieri-Kohlman VL, Pantilat SZ, Landefeld CS, Collard HR. Depression is a common and chronic comorbidity in patients with interstitial lung disease. Respirology. 2012 Apr;17(3):525-32. [CrossRef] [PubMed]
- Ryerson CJ, Berkeley J, Carrieri-Kohlman VL, Pantilat SZ, Landefeld CS, Collard HR. Depression and functional status are strongly associated with dyspnea in interstitial lung disease. Chest. 2011 Mar;139(3):609-616. [CrossRef] [PubMed]
- Wang C, Yang LS, Shi XH, Yang YF, Liu K, Liu RY. Depressive symptoms in aged Chinese patients with silicosis. Aging Ment Health. 2008 May;12(3):343-8. [CrossRef] [PubMed]
- Yildiz T, Eşsizoğlu A, Onal S, Ateş G, Akyildiz L, Yaşan A, Özmen CA, Cimrin AH. Quality of life, depression and anxiety in young male patients with silicosis due to denim sandblasting. Tuberk Toraks. 2011;59(2):120-5. [CrossRef] [PubMed]
- Diagnostic and Statistical Manual of Mental Disorders: DSM-5. American Psychiatric Association, 2013.
- Whooley MA, Avins AL, Miranda J, Browner WS. Case-finding instruments for depression. Two questions are as good as many. J Gen Intern Med. 1997 Jul;12(7):439-45. https://doi.org/10.1046/j.1525-1497.1997.00076.x [PubMed]
- Dawson SE, Madsen GE. Psychosocial and health impacts of uranium mining and milling on Navajo lands. Health Phys. 2011 Nov;101(5):618-25. [CrossRef] [PubMed]
- Mahler DA, Wells CK. Evaluation of clinical methods for rating dyspnea. Chest. 1988 Mar;93(3):580-6. [CrossRef] [PubMed]
- Arroll B, Goodyear-Smith F, Crengle S, Gunn J, Kerse N, Fishman T, Falloon K, Hatcher S. Validation of PHQ-2 and PHQ-9 to screen for major depression in the primary care population. Ann Fam Med. 2010 Jul-Aug;8(4):348-53. [CrossRef] [PubMed]
- Graham BL, Steenbruggen I, Miller MR, Barjaktarevic IZ, Cooper BG, Hall GL, Hallstrand TS, Kaminsky DA, McCarthy K, McCormack MC, Oropez CE, Rosenfeld M, Stanojevic S, Swanney MP, Thompson BR. Standardization of Spirometry 2019 Update. An Official American Thoracic Society and European Respiratory Society Technical Statement. Am J Respir Crit Care Med. 2019 Oct 15;200(8):e70-e88. [CrossRef] [PubMed]
- Hankinson JL, Odencrantz JR, Fedan KB. Spirometric reference values from a sample of the general U.S. population. Am J Respir Crit Care Med. 1999 Jan;159(1):179-87. [CrossRef] [PubMed]
- Radioactive Waste From Uranium Mining and Milling. 2 Mar. 2020, www.epa.gov/radtown/radioactive-waste-uranium-mining-and-milling.
- Bjørklund G, Christophersen OA, Chirumbolo S, Selinus O, Aaseth J. Recent aspects of uranium toxicology in medical geology. Environ Res. 2017 Jul;156:526-533. [CrossRef][PubMed]
- Yiin JH, Anderson JL, Daniels RD, Bertke SJ, Fleming DA, Tollerud DJ, Tseng CY, Chen PH, Waters KM. Mortality in a combined cohort of uranium enrichment workers. Am J Ind Med. 2017 Jan;60(1):96-108. [CrossRef] [PubMed]
- “Uranium Health Effects.” Uranium Health Effects, The Depleted UF6 Management Program Information Network, web.evs.anl.gov/uranium/guide/ucompound/health/index.cfm.
- Lestaevel P, Bussy C, Paquet F, Dhieux B, Clarençon D, Houpert P, Gourmelon P. Changes in sleep-wake cycle after chronic exposure to uranium in rats. Neurotoxicol Teratol. 2005 Nov-Dec;27(6):835-40. [CrossRef] [PubMed]
- Dawson SE, Madsen GE. American Indian uranium millworkers: a study of the perceived effects of occupational exposure. J Health Soc Policy. 1995;7(2):19-31. [CrossRef] [PubMed]
- Kreuzer M, Sogl M, Brüske I, Möhner M, Nowak D, Schnelzer M, Walsh L. Silica dust, radon and death from non-malignant respiratory diseases in German uranium miners. Occup Environ Med. 2013 Dec;70(12):869-75. [CrossRef] [PubMed]
- Shen CH, Chen HJ, Lin TY, Huang WY, Li TC, Kao CH. Association between pneumoconiosis and pulmonary emboli. A Nationwide Population-Based Study in Taiwan. Thromb Haemost. 2015 May;113(5):952-7. [CrossRef] [PubMed]
- Yu H, Zhang H, Wang Y, Cui X, Han J. Detection of lung cancer in patients with pneumoconiosis by fluorodeoxyglucose-positron emission tomography/computed tomography: four cases. Clin Imaging. 2013 Jul-Aug;37(4):769-71. [CrossRef] [PubMed]
- Graber JM, Stayner LT, Cohen RA, Conroy LM, Attfield MD. Respiratory disease mortality among US coal miners; results after 37 years of follow-up. Occup Environ Med. 2014 Jan;71(1):30-9. [CrossRef] [PubMed]
- Lu J, Jiang S, Ye S, Deng Y, Ma S, Li CP. Sequence analysis of the drug‑resistant rpoB gene in the Mycobacterium tuberculosis L‑form among patients with pneumoconiosis complicated by tuberculosis. Mol Med Rep. 2014 Apr;9(4):1325-30. [CrossRef] [PubMed]
- Go LH, Krefft SD, Cohen RA, Rose CS. Lung disease and coal mining: what pulmonologists need to know. Curr Opin Pulm Med. 2016 Mar;22(2):170-8. [CrossRef] [PubMed]
- Perini W, Snijder MB, Schene AH, Kunst AE. Prevalence of depressive symptoms among patients with a chronic nonspecific lung disease in five ethnic minority groups. Gen Hosp Psychiatry. 2015 Nov-Dec;37(6):513-7. [CrossRef] [PubMed]
- Boussoffara L, Boudawara N, Gharsallaoui Z, Sakka M, Knani J. Troubles anxiodépressifs et dilatation des bronches [Anxiety-depressive disorders and bronchiectasis]. Rev Mal Respir. 2014 Mar;31(3):230-6. French. [CrossRef] [PubMed]
- Kim Y, van Ryn M, Jensen RE, Griffin JM, Potosky A, Rowland J. Effects of gender and depressive symptoms on quality of life among colorectal and lung cancer patients and their family caregivers. Psychooncology. 2015 Jan;24(1):95-105. https://doi.org/10.1002/pon.3580 [PubMed]
- Guillien A, Laurent L, Soumagne T, Puyraveau M, Laplante JJ, Andujar P, Annesi-Maesano I, Roche N, Degano B, Dalphin JC. Anxiety and depression among dairy farmers: the impact of COPD. Int J Chron Obstruct Pulmon Dis. 2017 Dec 19;13:1-9. [CrossRef] [PubMed]
- Atlantis E, Fahey P, Cochrane B, Smith S. Bidirectional associations between clinically relevant depression or anxiety and COPD: a systematic review and meta-analysis. Chest. 2013 Sep;144(3):766-777. [CrossRef] [PubMed]
- Slatore CG, Harber P, Haggerty MC; American Thoracic Society Respiratory Impairment and Disability Evaluation Group. An official American Thoracic Society systematic review: Influence of psychosocial characteristics on workplace disability among workers with respiratory impairment. Am J Respir Crit Care Med. 2013 Nov 1;188(9):1147-60. [CrossRef] [PubMed]
- Salk RH, Hyde JS, Abramson LY. Gender differences in depression in representative national samples: Meta-analyses of diagnoses and symptoms. Psychol Bull. 2017 Aug;143(8):783-822. [CrossRef] [PubMed]
- Csordas TJ, Storck MJ, Strauss M. Diagnosis and distress in Navajo healing. J Nerv Ment Dis. 2008 Aug;196(8):585-96. [CrossRef] [PubMed]
- Ka'apu K, Burnette CE. A Culturally Informed Systematic Review of Mental Health Disparities Among Adult Indigenous Men and Women of the USA: What is known? Br J Soc Work. 2019 Jun;49(4):880-898. [CrossRef] [PubMed]
- Mills SD, Fox RS, Pan TM, Malcarne VL, Roesch SC, Sadler GR. Psychometric Evaluation of the Patient Health Questionnaire-4 in Hispanic Americans. Hisp J Behav Sci. 2015 Nov;37(4):560-571. [CrossRef] [PubMed]
- Rizvi SJ, Pizzagalli DA, Sproule BA, Kennedy SH. Assessing anhedonia in depression: Potentials and pitfalls. Neurosci Biobehav Rev. 2016 Jun;65:21-35. [CrossRef] [PubMed]
Cite as: Sharma S, Shore XW, Mohite S, Myers O, Kesler D, Vlahovich K, Sood A. Association between Spirometric Parameters and Depressive Symptoms in New Mexico Uranium Workers. Southwest J Pulm Crit Care. 2021;21(2):58-68. doi: https://doi.org/10.13175/swjpcc015-20 PDF











